
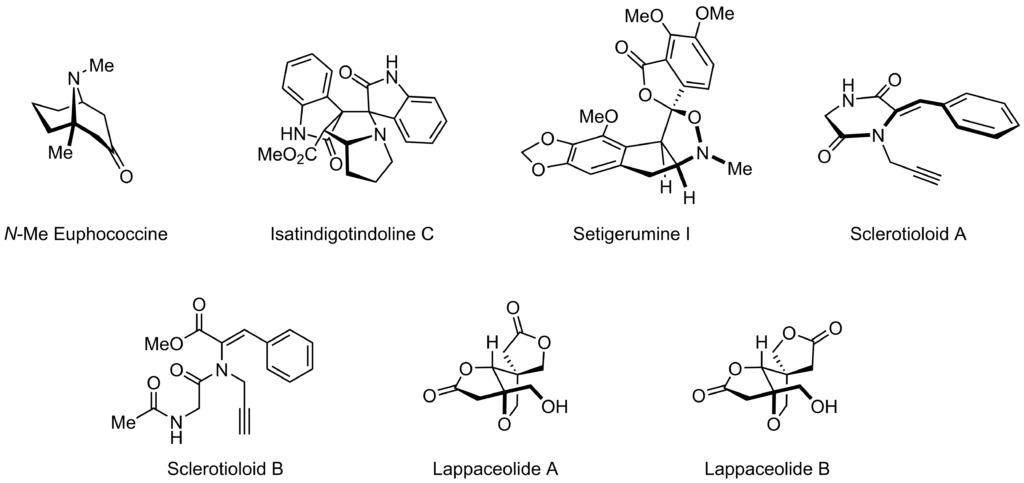
The ETOS group operates at the interface between experimental and theoretical organic synthesis. Our work is largely driven by inspiration drawn from polycylic natural products which are used as platforms for synthetic strategy design, development of new synthetic methods, and the study of mechanistically intriguing processes.
The overarching theme in all of our research efforts is the fundamental physical understanding of the chemical processes involved. With deeper understanding of the underlying regio- and stereoselectivity elements comes greater control over the chemistry at hand. In the long run, we hope such an approach of combining synthesis with computational tools on a fundamental level will provide chemists with reliable tools for a-priori prediction even in the realm of dauntingly complex natural product syntheses.
Our labs are fully facilitated for modern organic synthesis research (Schlenk lines, SPSS solvent drying system, µW reactors, NextGen 300+ CombiFlash unit) and Aalto University provides us with large-scale instrumentation (400 and 600 MHz Bruker NMRs, LC/q-TOF MS). We also have access to the local Aalto computer clusters, and especially the mylly1 server (AMD EPYC 7702, 128 cores) with orca, gaussian, xtb, and crest available for routine use.
Organic synthesis
Organic synthetic reactions should be readily accessible to the entire scientific community, allowing people to easily make the molecules they need. This is why we are interested in the development of reliable bench-stable reagents, and spot-to-spot open-flask synthetic methods. To foster new synthetic ideas and to truly acid-test the limitations of current chemistry, we are pursuing the development of ideal syntheses for polycylic natural products. In particular, we are interested in the synthesis and biochemistry of structurally unique racemic alkaloids.
Relevant publications:
- Pallerla, R. R.; Hakola, J.; Härkönen, L.; Siitonen, J. H.* “Biomimetic Total Synthesis of (±)-Lappaceolides A and B”, Org. Lett. 2025, ASAP (DOI).
- Karta, M.; Pallerla, R. R.; Siitonen, J. H.* “Divergent Two-Step Total Synthesis of Sclerotioloid A and B”, Synlett, 2025, 36, A–D (DOI).
- Serna, A. V.; Kürti, L.*; Siitonen, J. H.* “Total Synthesis of (±)-Setigerumine I: Biosynthetic Origins of the Elusive Racemic Papaveracaea Isoxazolidine Alkaloids”, Angew. Chem. Int. Ed. 2021, 60, 27236 (DOI).
- Siitonen, J. H.*; Lira, S.; Youssufuddin, M.; Kürti, L.* “Total Synthesis of Isatindigotindoline C”, Org. Biomol. Chem. 2020, 18, 2051 (DOI). Featured in Org. Chem. Highlights: Alkaloid Synthesis
Theory and computation
How can we mathematically represent organic synthesis and what insights does this give us? We are developing algorithms, data structures and mathematical representations, especially those based on graph theory, to provide new ways of looking at organic synthesis.
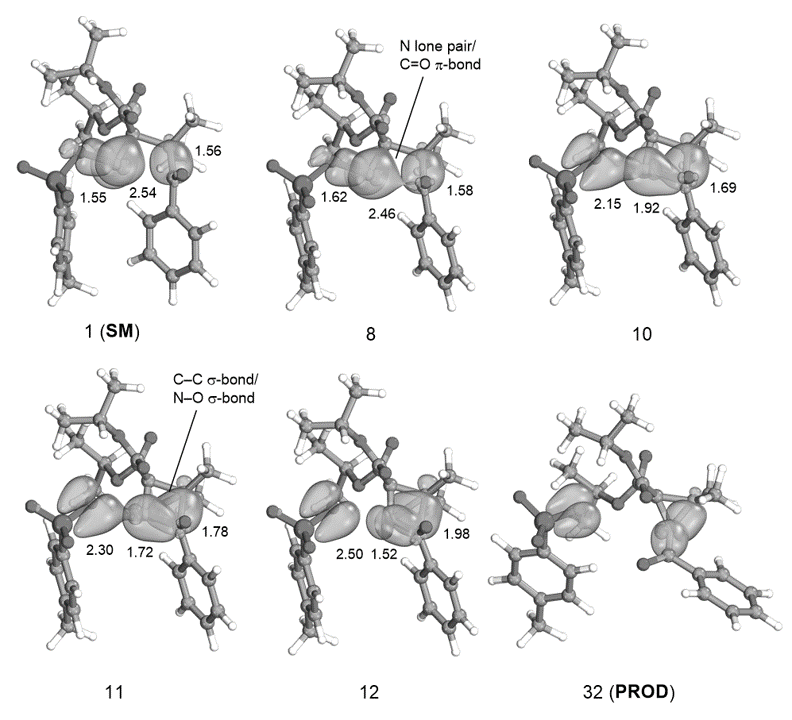
In addition, we are heavily involved in studying reaction mechanisms using theoretical methods. Main quantum-chemistry tools used are Gaussian, xTB, crest, ORCA, Molcas, NCIplot and IboView. In addition, we are applying Schrödinger Maestro for visualization needs. For studying energy landscapes a breadth of tools, including NEB and GSM are used. We are also currently expanding into multiconfigurational methods. In particular, we are focused on applying intrinsic bonding orbitals (IBOs) in combination with minimum energy pathway finding methods as tools to understand the inner workings of chemical reactions.
Relevant publications:
- Kattamuri, P. V.; Zhao, J.; Das, T. K.; Siitonen, J. H.*; Morgan, N. Ess, D. H.*, Kürti, L.* “Aza-Quasi-Favorskii Reaction: Construction of Highly Substituted Aziridines through a Concerted Multi-bond Rearrangement Process”, J. Am. Chem. Soc. 2022, 144, 24, 10943 (DOI).
- Csókás, D.; Siitonen, J. H.; Pihko, P. M.*; Papai, I.* “Conformationally Locked Pyramidality Explains the Methylation Stereochemistry of trans-Fused Butyrolactones”, Org. Lett. 2020, 22, 12, 4597 (DOI).
- Cheng, Q.-Q.; Zhou, Z.; Jiang, H.; Siitonen, J. H.; Ess, D. H.*; Zhang X.; Kürti, L.* “Organocatalytic nitrogen-transfer to unactivated olefins via transient oxaziridines”, Nature Catalysis 2020, 3, 386. (DOI).
Chemistry education
Outside of organic chemistry research one of our main efforts in chemistry education is aimed at helping and encouraging kids interested who are interested in science. We want to help kids who are interested in science get the support they need to become the next generation of scientist and engineers.
We are also interested in providing university level students with a comprehensive understanding of chemistry. Chemistry concepts should not be divided into separate categories such as physical, inorganic, organic or analytical, but rather they should be unified to form a coherent body of information. The same underlying physics, quantum mechanics and thermodynamics, governs all of chemistry.
As for learning theories and pedagogy, we are studying the effects of emotions on learning science. Oftentimes sciences are seen as cold and separate from emotions. However, emotions play a key role in science education and should be taken into account when planning, executing and evaluating truly inclusive higher education.
Relevant publications:
- Nurmi, T.*; Siitonen, J. H.* “Upper Secondary School and University Level Students’ Perceptions of Extractions in Context: Experiences from a Simple Laboratory Experiment”, ChemRxiv preprint, 2021.
Funding



